Resources
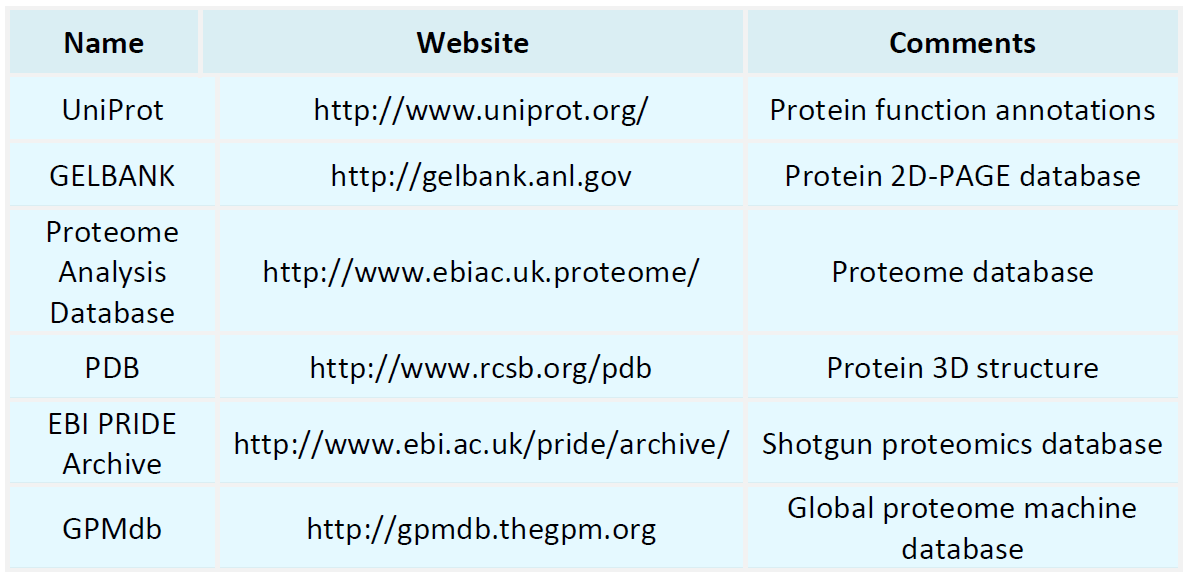
Proteomics Databases
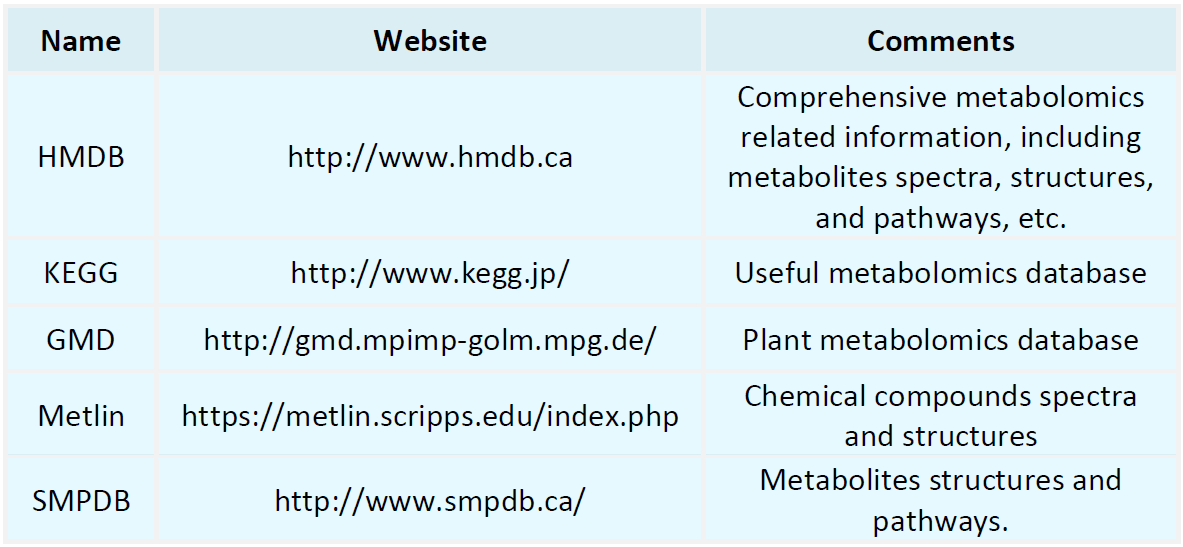
Metabolomics Databases
-
• MALDI-TOF for protein identification
MALDI TOF for protein identification is a technique that combines matrix-assisted laser desorption/ionization (MALDI) with time-of-flight mass spectrometry (TOF). In proteomics research, understanding and identifying the structure and function of proteins is essential. MALDI TOF for protein identification determines the composition and structural information of proteins by analyzing their mass-to-charge ratio (m/z), with the key advantage of enabling accurate mass measurement without compromising prot......
-
• MALDI-TOF Mass Spectrometry for Microorganism Identification
MALDI-TOF mass spectrometry for microorganism identification is a technique that utilizes matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) to identify and classify microorganisms. The core principle involves desorbing and ionizing protein molecules in a sample using laser energy, followed by determining their mass-to-charge ratio (m/z) based on their time of flight. This process generates characteristic fingerprint spectra that represent the protein compositi......
-
• What Determines the Isoelectric Point of Proteins
The isoelectric point of proteins (pI) is the pH at which a protein has no net charge, meaning it is electrically neutral. Understanding the isoelectric point of proteins is essential for various biochemical processes, including protein purification and understanding protein behavior in different pH environments. The isoelectric point is determined by factors such as the amino acid composition of the protein and the pKa values of individual amino acids. Amino Acid Composition The isoelectric point o......
-
• In Silico Drug Target Identification
In silico drug target identification employs advanced computational techniques to predict and discover molecular structures or sites within organisms that are likely to interact with pharmaceuticals. This approach relies on precise three-dimensional modeling of biological macromolecules, enabling researchers to simulate molecular interactions and predict potential binding sites. Common methods involved in this process include molecular dynamics simulations, binding free energy calculations, and molecu......
-
• Processing Proteomics Data with R
Processing proteomics data is an important aspect of bioinformatics, and R programming language is widely used due to its powerful capabilities in statistical computation and graphical visualization. In proteomics data analysis, R can be applied to a variety of tasks, including but not limited to data preprocessing, mass spectrometry data analysis, protein identification, quantification, biomarker discovery, as well as functional and pathway analysis. Below are some R packages and resources that may b......
-
Oligonucleotides, including deoxyribonucleic acid (DNA) and ribonucleic acid (RNA), are increasingly being utilized in diagnostics and therapeutics. For instance, DNA can be introduced into immune cells to genetically modify them, enabling the expression of chimeric antigen receptor (CAR) proteins for cell-based immunotherapy. Various types of RNA, including messenger RNA (mRNA) and small interfering RNA (siRNA), are also employed for transient protein expression and protein expression interference, r......
-
• How the Coefficient of Variation (CV) Is Calculated in Proteomics
In proteomics, the coefficient of variation (CV) is a statistical metric used to quantify the degree of variation in repeated measurements of protein expression levels. CV Calculation Formula The CV is calculated using the following formula: CV = (σ / μ) × 100% where σ represents the standard deviation of the dataset, and μ denotes the mean value. The CV is expressed as a percentage and reflects the extent of dispersion relative to the mean. A lower CV indicates less variation, suggesting higher r......
-
• Proteomics: From Quantification to Functional Insights
Basic Concepts of Proteomics Proteomics is a crucial branch of biology that investigates all the proteins within an organism, including their structure, function, and interactions. These proteins constitute the organism's "proteome," and they are the main executors of biological activities. Quantitative Analysis in Proteomics Quantitative proteomics is an essential field within proteomics that focuses on accurately measuring the relative or absolute abundance of each protein in a cell. With the aid ......
-
• A Detailed Workflow of TMT-Based Quantitative Proteomics
TMT-based quantitative proteomics is an advanced technique employed for the quantitative analysis of protein expression. This method utilizes specific chemical tags to label protein samples, enabling comparative analysis of protein expression across different samples via mass spectrometry. The standard experimental workflow for TMT-based quantitative proteomics comprises the following steps: 1. Sample Preparation (1) Protein Extraction: Proteins are extracted from cells, tissues, or biological fluid......
-
• Methods for Protein Content Determination
Protein content determination is a significant research topic in the fields of biochemistry and food science. There are various methods available to determine the protein content in samples, each with its specific application scenarios and advantages and disadvantages. Below is a detailed introduction to several common methods for protein content determination: Kjeldahl Method 1. Principle The Kjeldahl method is a classic technique for protein content determination. It estimates protein content indi......
How to order?







