Resources
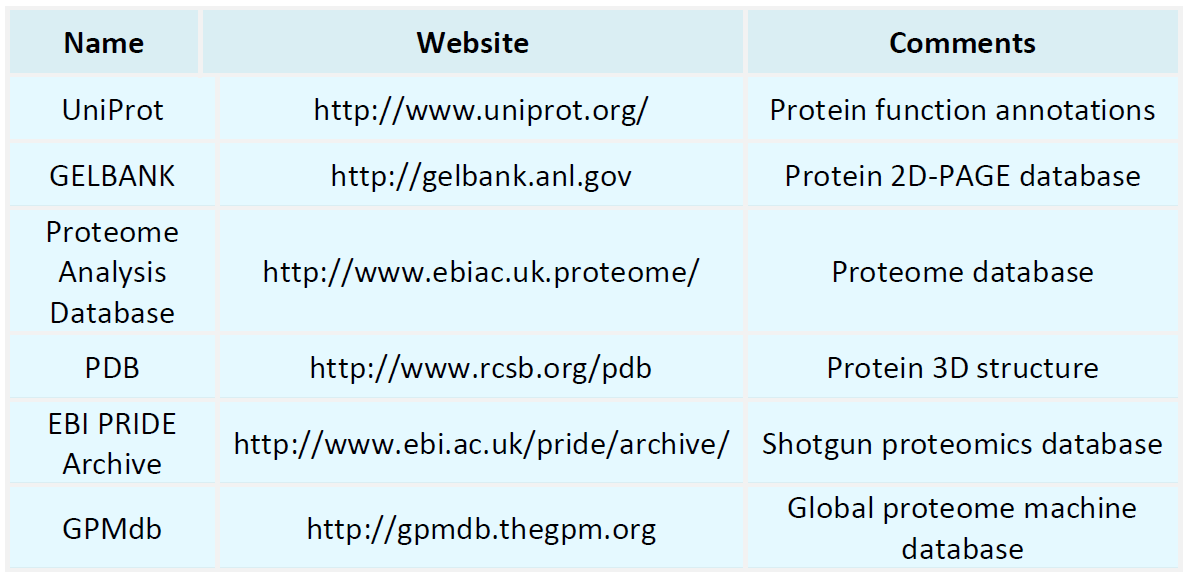
Proteomics Databases
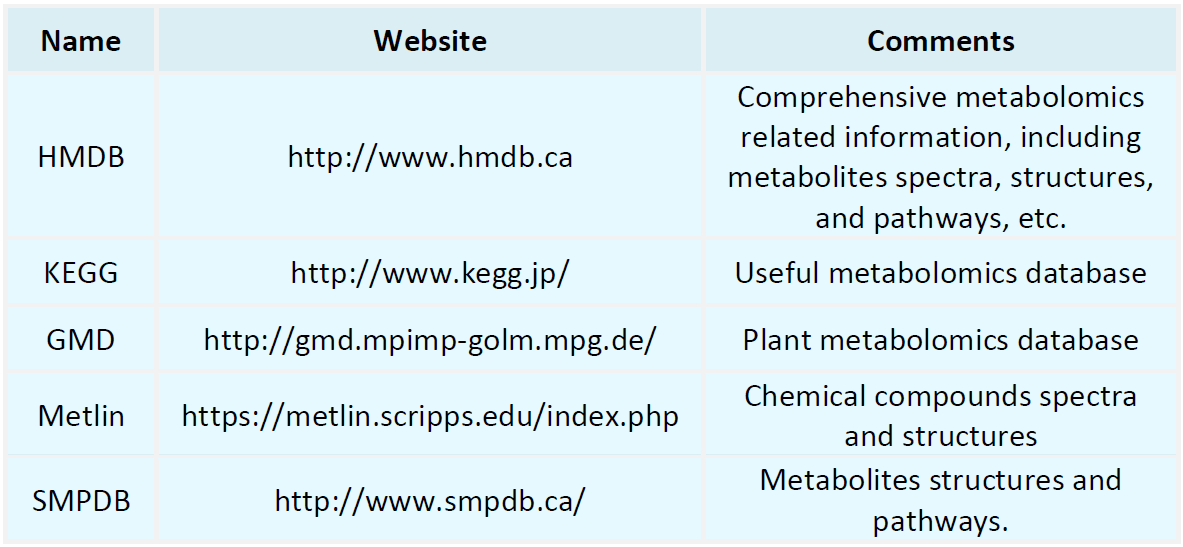
Metabolomics Databases
-
• How to Improve Sensitivity in Targeted PTM Detection by Mass Spectrometry?
Protein post-translational modifications (PTMs), including phosphorylation, acetylation, ubiquitination, and glycosylation, play pivotal roles in regulating signal transduction, cellular metabolism, and disease pathogenesis. Relative to unmodified proteins, PTM-bearing proteins/peptides are frequently low in abundance, exhibit heterogeneous modification sites, and may be chemically or enzymatically unstable; consequently, achieving high sensitivity and specificity remains a central challenge. Owing to......
-
• Why Is Targeted PTM Analysis Essential in Proteomics Research?
In modern life-science research, proteomics has evolved from cataloguing proteins toward elucidating how proteins are regulated and how they shape cellular states. A central theme in this transition is the systematic characterization of post-translational modifications (PTMs). Across basic biology, studies of disease mechanisms, and drug-target discovery, targeted PTM analysis (targeted PTM proteomics) has become an indispensable approach. This article explains why targeted PTM analysis is a major foc......
-
• How to Validate Exosome Integrity After Purification?
Exosomes are nanoscale vesicles generated through the endosomal pathway and are widely present in various biological fluids, including plasma, urine, saliva, and milk. They have attracted extensive interest due to their important roles in intercellular communication, disease biomarker discovery, and drug delivery. However, functional investigations of exosomes rely fundamentally on the preservation of their structural and biological integrity. Loss of integrity may introduce experimental bias and even......
-
• How to Improve Exosome Purity Without Losing Yield?
Exosomes are key mediators of intercellular communication and have, in recent years, demonstrated substantial potential in tumor biology, neurodegenerative diseases, liquid biopsy, and drug delivery. However, exosomal functions and associated analytical readouts can be readily confounded by contaminants, including free proteins, liposomes, cell debris, and other non-specific components, thereby creating major obstacles for downstream omics analyses, functional validation, and clinical translation. In ......
-
• How to Purify Exosomes Effectively: Comparing Ultracentrifugation, SEC, and Immunoaffinity Methods
Exosomes are small vesicles released by diverse cell types and are commonly found in plasma, urine, saliva, milk, and cell-culture supernatants. Serving as key mediators of intercellular communication, exosomes have attracted sustained interest from both academia and industry in recent years, particularly for early cancer detection, disease biomarker discovery, and the development of delivery systems. Nevertheless, because exosomes are small (30–150 nm) and share highly overlapping physical characteri......
-
• Glycoproteomics in Drug Development
With the rapid advancement of precision medicine and the biopharmaceutical industry, glycoproteomics, as a cutting-edge technology for the systematic investigation of protein glycosylation, has emerged as an integral component of modern drug development. Its broad applications in target identification, biomarker discovery, and quality control of biopharmaceutical products provide high-resolution molecular insights that substantially facilitate innovative drug research and development. Overview of Gly......
-
• Classification of Protein Glycosylation: An Overview
Protein glycosylation is one of the most common and structurally complex post-translational modifications (PTMs), widely distributed in eukaryotes and also present in certain prokaryotes. It plays critical roles in regulating protein folding, stability, and subcellular localization, and is extensively involved in diverse biological processes such as cell recognition, signal transduction, and immune regulation. Under pathological conditions, including cancer, autoimmune diseases, and metabolic syndrome......
-
Functional annotation and signaling pathway analysis of acylated proteins are essential for understanding their roles in cellular processes, metabolic regulation, and disease mechanisms. What Are Acylated Proteins? Why Should They Be Studied? Acylation represents an important class of post-translational protein modifications, with common types including: Acetylation Propionylation Butyrylation Succinylation Malonylation Palmitoylation, among others. These modifications typically occur o......
-
• How to Evaluate the Quality of Custom Synthetic Peptides?
In experimental applications such as antibody generation, protein–protein interaction studies, mass spectrometry–based analysis, and vaccine development, the quality of custom synthetic peptides directly determines the reliability and reproducibility of experimental outcomes. With the expanding use of peptides in functional studies, molecular targeting, and novel drug discovery, increasing attention has been devoted to critical parameters including purity, sequence accuracy, and stability. In particul......
-
In the post-genomic era, post-translational modifications (PTMs) have attracted increasing attention as critical regulatory mechanisms underlying protein functional diversity. Among these, acylation, particularly lysine acetylation, propionylation, butyrylation, and malonylation, represents an important class of lysine modifications that are extensively involved in the regulation of key biological processes, including metabolism, signal transduction, and chromatin remodeling. However, these modificati......
How to order?







